| 論文篇名 | 英文:MTHFR C677T polymorphism increases MTX sensitivity via the inhibition of S-adenosylmethionine and de novo purine synthesis 中文:亞甲基四氫葉酸C677T基因多態性透過抑制S-腺苷甲硫氨酸和嘌呤合成提高疾病修飾抗風濕病藥物敏感性 |
| 期刊名稱 | CLINICAL SCIENCE |
| 發表年份,卷數,起迄頁數 | 2019, 133(2), 253-267 |
| 作者 | Wang, Yi-Cheng;Wu, Ming-Tsung;Tang, Feng-Yao;Chen, Der-Yuan;Ko, Hsin-An;Shane, Barry;Huang, Wen-Nan;Chiang, En-Pei Isabel (蔣恩沛)* |
| DOI | 10.1042/CS20180932 |
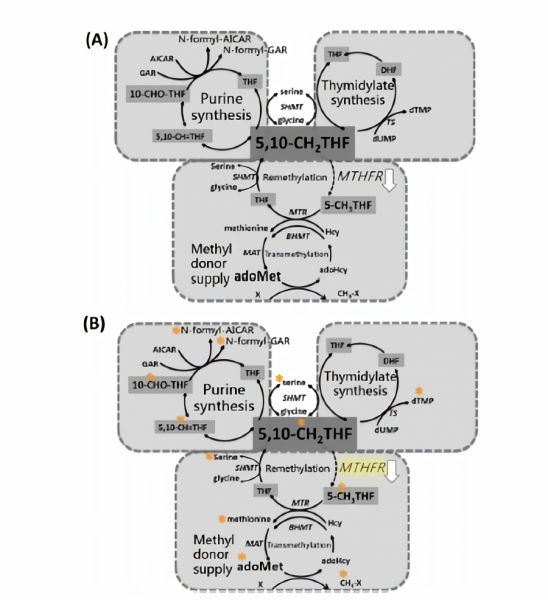
| 中文摘要 | 實驗目的 對於類風溼性關節炎患者給予的疾病修飾抗風濕病藥物治療之基因組學上,目前對於葉酸代謝基因變異之影響尚無長期明確方針。我們想探討在給予葉酸拮抗劑 methotrexate (MTX) 情況下,葉酸代謝基因methylenetetrahydrofolate reductase (MTHFR) 會如何影響細胞整體代謝動力學。實驗設計 我們建立兩種不同細胞模型: (1) 利用病毒轉染的方式將shRNA 送入到 HepG2 中將其 MTHFR 進行穩定抑制 (2) 以 Epstein-Barr virus 轉染人類淋巴母細胞表達MTHFR 677C及 677T不同基因型態的淋巴母細胞。進行橫斷面研究比較具有MTHFR-677CC、 CT和TT 使用疾病修飾抗風濕病藥藥物之類風濕性關節炎患者在其疾病活性和用藥劑量(n = 120)。結果 與MTHFR-CC相比,MTHFR-TT攜帶者具有較低的每週MTX劑量(9.8 ± 3.3 v.s 12.1 ± 3.5,p <0.05)。與 MTHFR-677CC (32 / 57,56%)和MTHFR-677CT(30 / 51,59%)相比有較多MTHFR-TT攜帶者(8 / 11,73%)發生MTX副作用。其他非葉酸拮抗劑疾病修飾抗風濕病藥藥物則沒有發現基因型上的差異。在給予相同劑量的 MTX 淋巴母細胞對於其存活會更敏感,然而在HepG2 細胞中則會合成蛋白與胸腺嘧啶抑制S-腺苷甲硫氨酸生合成。在帶有MTHFR-C677T 的淋巴母細胞中,有無給予 MTX 會改變蛋白轉換率及葉酸調控單碳代謝的分流。在HepG2 細胞中給予MTX,MTHFR 會顯著影響細胞轉甲基分流及S-腺苷甲硫氨酸動態平衡但並不影響細胞核甘酸的生合成。總結 本研究結合細胞模型、穩定同位素標定及動力學探討及人類基因型態如何影響藥物反應,研究更進一步發現在給予 MTX 治療上 MTHFR 基因變異如何影響肝中轉甲機的動態平衡。我們提供此平台將有助於預測抗葉酸藥物的遺傳影響,並進一步描述疾病修飾抗風濕病藥物治療中的組織特異性標靶途徑。我們也建議在臨床實踐中應考慮遺傳因素之影響。 關鍵字: 代謝; 免疫抑製劑; 藥物基因組學; 類風濕性關節炎; 單核苷酸多態。 |
| 英文摘要 | Objective: Currently, no guidelines are established for pharmacogenomic testing involving folate metabolic genes in long-term disease-modifying antirheumatic drugs' (DMARD) therapies. We carefully investigated how common genetic variations in methylenetetrahydrofolate reductase (MTHFR) influence cellular metabolic kinetics in response to methotrexate (MTX). Designs: Two distinct cell models: HepG2 with stabilized MTHFR inhibition using shRNA delivered by a Lentiviral vector; and Epstein-Barr virus transformed human lymphoblasts expressing MTHFR polymorphic allele 677C and 677T were used. Disease activity and DMARD use were compared between MTHFR-677CC, CT and TT rheumatoid arthritis (RA) patients in a cross-sectional study (n= 120). Results: Compared with MTHFR-CC, MTHFR-TT carriers had lower mean weakly MTX dose (9.8 /- 3.3 compared with 12.1 /- 3.5, P< 0.05). More MTHFR-TT carriers (8/11, 73%) reported MTX-related side effects compared with MTHFR-677CC (32/57, 56%) and MTHFR-677CT (30/51, 59%). No genotypic difference was found in other DMARDs. At the same dose of MTX, lymphoblasts were more sensitive in cell survival, protein and thymidine syntheses whereas HepG2modelsweremore susceptible to the inhibition of S-adenosylmethionine (adoMet) synthesis. MTHFR-C677T altered protein turnover and folate mediated 1-carbon metabolic fluxes in lymphoblasts with and withoutMTX. MTHFR function significantly affected transmethylation fluxes and adoMet homeostasis but not nucleotide biosyntheses in MTX-treated HepG2 cell-lines. Conclusion: Combining cellmodels, kinetic studies, and genetic tests in humans, the present study gives insight on how MTHFR effects hepatic transmethylation homeostasis during MTX therapy. We provide platforms that help predict the genetic impact on antifolate drugs, and further delineate tissue-specific target pathway in DMARD therapies. We suggest that genetic factors should be taken into account in clinical practice. |
【學術亮點】亞甲基四氫葉酸C677T基因多態性透過抑制S-腺苷甲硫氨酸和嘌呤合成提高疾病修飾抗風濕病藥物敏感性 2019-01-03
循環農業:農業廢棄物再資源化【食生系蔣恩沛特聘教授】






